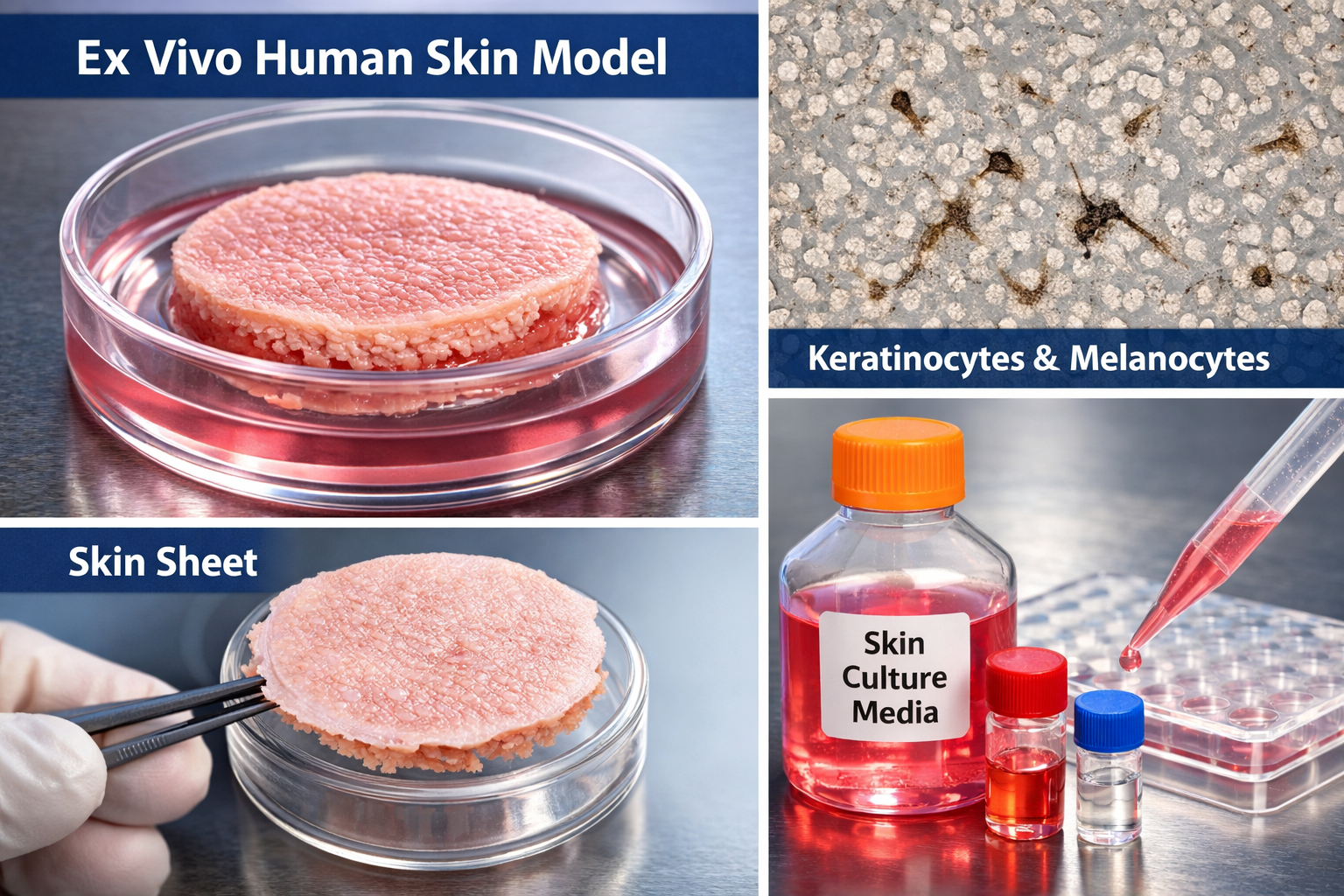
SKIN TISSUES and media
1 Ex. Vivo Human Skin Model (OECD 428 compliance)
2 Full-Thickness Skin Sheet (with or without Adipose tissues)
3 Keratinocytes and Melonocytes cells
4 Skin cells Culture Media

Ex
Vivo Human Skin Models
At
NeoJenBio, we are redefining how skin absorption and safety studies are
performed—by bringing research closer to real human biology.
Our Ex Vivo Human Skin Tissues, combined with optimized skin culture media, provide a robust and predictive solution for skin absorption assays compliant with OECD 428. Designed to support modern toxicology and preclinical research, our platforms help generate reliable, human-relevant data while meeting regulatory expectations.
Our Ex Vivo Human Skin Tissues, combined with optimized skin culture media, provide a robust and predictive solution for skin absorption assays compliant with OECD 428. Designed to support modern toxicology and preclinical research, our platforms help generate reliable, human-relevant data while meeting regulatory expectations.
🔬 Why choose NeoJenBio skin solutions?
🧬 Human-based skin systems
Developed to closely replicate physiological skin responses, ideal for skin absorption and permeability studies under OECD 428 guidelines.
🧪 Optimized skin culture media
Carefully formulated to preserve tissue stability and integrity, ensuring consistency, reproducibility, and experimental reliability throughout your studies.
🌍 Ethical & regulatory alignment
Ethically sourced human tissues, rigorous quality control processes, and full alignment with international regulatory standards—supporting the transition away from animal testing.
🧬 Human-based skin systems
Developed to closely replicate physiological skin responses, ideal for skin absorption and permeability studies under OECD 428 guidelines.
🧪 Optimized skin culture media
Carefully formulated to preserve tissue stability and integrity, ensuring consistency, reproducibility, and experimental reliability throughout your studies.
🌍 Ethical & regulatory alignment
Ethically sourced human tissues, rigorous quality control processes, and full alignment with international regulatory standards—supporting the transition away from animal testing.
OECD 428 – Skin Absorption: In Vitro Method
